The Kengaku Group
KUIAS iCeMS, Kyoto University
Research
Research outline
The cerebellar cortex and hippocampus are formed of clear layered cytoarchitectures that provide favorable systems for the study of the formation and function of neural circuitry. We have developed in vitro reconstructions of cell morphogenesis and migration during cerebellar and hippocampal development. We aim to discover novel phenomena and the underlying rules of brain formation using multidisciplinary approaches, including advanced microscopies, cell and molecular biology, and mechanobiology. Our main research directions can be summarized under the following three themes:
- Mechanisms and principles of branch pattern formation of dendrites
- Cellular and molecular dynamics of neuronal migration
- New techniques based on light microscopy
研究内容
小脳皮質と海馬は、大脳皮質に比較すると構造が単純かつ明瞭で、神経回路について生理学的・解剖学的知見が蓄積しています。我々は小脳皮質と海馬を主要なモデル系として発生過程の諸現象を培養下で再構成し、高解像ライブイメージング技術などを用い、以下3つの項目について研究を行っています。
Mechanisms and principles of branch pattern formation of dendrites
Dendrite morphologies of CNS neurons are highly diverse, depending on cell type and function. The architecture of dendritic arbors critically affects the integration of neuronal inputs and propagation of chemical signals, and hence determines the connectivity of neurons. The question of how neurons acquire their appropriate morphology is a major issue in the study of neuronal development. In spite of the increasing number of molecular signals that have been identified as regulators of dendritic arborization patterns, the precise function of each molecule in the specific steps of branch dynamics largely remains elusive.
Cerebellar Purkinje cells develop intricate dendritic arbors with minimal branch overlap. We developed a method of long-term time-lapse observation of dendritic branch dynamics in growing Purkinje cells in culture. Using a combinatorial approach with quantitative image analyses and computer-aided simulation, we identified the fundamental rules of growth dynamics that govern the construction of the characteristic dendritic patterns in Purkinje cells(Fujishima et al., Development, 2012). We also demonstrated that a small change in actin dynamics led to a siginifant difference in dendrite growth dynamics and the untimate branch pattern in a mature neuron (Kawabata Galbraith et al., Cell Reports, 2018). Purkinje cells arborize fan-shaped dendrites which form perpendicular contacts with the afferent granule cell axons. We developed a new culture system to reconstitute the axon-dendrite wiring topology. Using this, we identified molecular players involved in the perpendicular connectivity (Fujishima et al., 2020).
Time-lapse observation and computer-assisted simulation of dendrite formation in cultured Purkinje cells
培養プルキンエ細胞の樹状突起形成ダイナミクスとシミュレーション
長期タイムラプス観察により樹状突起形成過程を解析し、必要最小限のパラメータを抽出して モデル細胞を再構築した。
We have successfully visualized the dynamic motility of organelles, including the nucleus, centrosomes, mitochondria and Golgi apparatus, in developing neurons. We are investigating how the neural activity during circuit formation affects dendrite arborization.
Super-resolutioin images of mitochondria dynamics in cultured hippocampus neuron
培養した海馬ニューロンのミトコンドリア動態の超解像イメージ
培養した海馬ニューロンの細胞質(緑)とミトコンドリア(マゼンダ、白)の動態を超解像顕微鏡(Zeiss Airy scan)を用いて観察した。
樹状突起パターン形成のダイナミクスと原理の解明
中枢神経系ニューロン樹状突起の分岐パターンは、シナプス結合する入力線維の種類と数を決め、また膜電位と細胞内シグナルの拡散に影響してニューロンの情報処理特性を規定します。このためニューロンは機能に応じて個性的な分岐パターンを獲得します。しかし発達中の脳で数日から数週間かけて完成する樹状突起のパターン形成過程を長期間観察するのは技術的に困難で、ダイナミクス(伸長・分岐・退縮)の組合せにより細胞種毎に異なる分岐パターンが獲得される機構は明らかでありません。
我々は中枢神経系ニューロンの中でも際立って緻密な樹状突起を形成する小脳プルキンエ細胞を用い、分散培養下で樹状突起発達過程を1週間以上連続観察する系を確立し、樹状突起ダイナミクスの定量的解析と数理解析を用いてその形成原理を明らかにしました(Fujishima et al. 2012)。また、分化中の細胞骨格アクチン動態のわずかなズレが樹状突起ダイナミクスに影響し、成熟したニューロンの分岐の形を大きく変えてしまうことを証明しました(Kawabata Galbraith et al. 2018;詳細はこちら)。プルキンエ細胞は脳内で入力する顆粒細胞軸索と直交する扇型の樹状突起を展開します。神経回路トポロジーを解析する新たな培養再構成系を開発し、プルキンエ細胞樹状突起が顆粒細胞軸索に対し垂直方向に伸展することを観察し、その分子機構の一端を明らかにしました(Fujishima 2020)。
発達中の樹状突起内でゴルジ体、ミトコンドリアなどのオルガネラの動態を可視化することにも成功しています。現在、神経活動が樹状突起発達に与える影響とその分子機構を解析しています。
Reference
Fujishima K, Kurisu J, Yamada M, Kengaku M. (2020) βIII spectrin controls the planarity of Purkinje cell dendrites by modulating perpendicular axon-dendrite interactions. Development 16;147(24):dev194530. doi: 10.1242/dev.194530.
Kawabata-Galbraith, K., Fujishima, K., Mizuno, H., Lee, S.J., Uemura, T., Sakimura, K., Mishina, M., Watanabe, N. and Kengaku, M. (2018) MTSS1 regulation of actin-nucleating formin DAAM1 in dendritic filopodia determines final dendritic configuration of Purkinje cells. Cell Rep. 24(1):95-106.
Hatsukano, T., Kurisu, J., Fukumitsu, K., Fujishima, K. and Kengaku, M. (2017) Thyroid hormone induces PGC-1 α during dendritic outgrowth in mouse cerebellar Purkinje cells. Front Cell Neurosci. 11:133
Fukumitsu, K., Hatsukano, T., Yoshimura, A., Heuser, J., Fujishima, K. and Kengaku, M. (2016) Mitochondrial fission protein Drp1 regulates mitochondrial transport and dendritic arborization in cerebellar Purkinje cells. Mol Cell Neurosci. 71:56-65
Fukumitsu, K., Fujishima, K., Yoshimura, A., Wu, Y.K., Heuser, J. and Kengaku, M. (2015) Synergistic action of dendritic mitochondria and creatine kinase maintains ATP homeostasis and actin dynamics in growing neuronal dendrites. J. Neurosci. 35(14):5707- 5723.
Wu, Y.K., Fujishima, K. and Kengaku, M. (2015) Differentiation of Apical and Basal Dendrites in Pyramidal Cells and Granule Cells in Dissociated Hippocampal Cultures. PLoS ONE 10(2) e0118482.
Fujishima, K., Horie, R., Mochizuki, A. and Kengaku M. (2012) Principles of branch dynamics governing shape characteristics of cerebellar Purkinje cell dendrites. Development 139: 3442-3455.
Cellular and molecular dynamics of neuronal migration
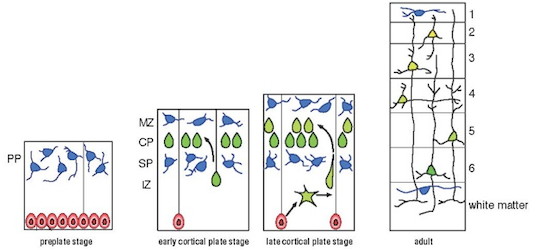
Neurons are generated from neural stem cells in the germinal layer, and then migrate through the crowded neural tissues toward their specific sites of function within the cortex. Failure in neuronal migration may cause severe brain malformation and psychiatric disorders.
Cerebellar granule cells are the excitatory interneurons of the cerebellar cortex, which undergo significant migration during cortex formation. We have established a time-lapse imaging system for quantitative analyses of granule cell migration in organotypic cultures, which retain the cell architecture and environment of the cerebellar cortex. We have successfully visualized organelle dynamics in migrating neurons in the brain tissue and have proposed a novel model for neuronal migration (Umeshima et al., 2007, Umeshima et l., 2012).
Neuronal migration in cultured cerebellar tissue
小脳皮質を移動する顆粒細胞
電気穿孔法で小脳皮質の顆粒細胞前駆細胞に蛍光分子を発現させ、切片培養下で分化後の顆粒細胞移動を観察した。
We have recently developed an in vitro system for analyses of motion dynamics of neuronal migration at a high spatio-temporal resolution using spinning-disc confocal microscopy. We have found that the migrating nucleus exhibits highly dynamic motion, including sharp deformation and rotation, suggesting the involvement of multiple motor systems (Wu et al., 2018). We now seek to visualize the force which drives migration, by quantitative measurement of the rheological properties of migrating neurons.
ニューロン移動のダイナミクス制御の分子・力学機構
ニューロンとグリアの前駆細胞である神経上皮細胞は、脳室帯と呼ばれる分裂層で活発に分裂して数を増やします。最終分裂して分化したニューロンは、脳表面に向って細胞移動し、順に積み上がって皮質を形成します(図)。ニューロン移動の異常は層構造の乱れや神経核の欠失による回路の配線ミスを招き、てんかんや精神遅滞、運動失調など様々な神経疾患を引き起こします。
小脳皮質の介在ニューロンである顆粒細胞は、発生過程で最も大規模に移動する細胞のひとつです。我々は生後発達中の小脳組織を器官培養し、皮質内を移動する顆粒細胞の動きを長時間観察するリアルタイムイメージング系を確立しています。このシステムを用い、組織内を移動するニューロンのオルガネラのダイナミクスの観察に世界に先駆けて成功しました(Umeshima et al., 2007、Umeshima et al., 2012)。スピニングディスク型共焦点顕微鏡を用いてさらに時空間解像度を上げて観察すると、ニューロン核は前進のみならず回転や変形など、複数のモーター分子活性を示唆する複雑な動きを伴うことが明らかとなりました(Wu et al., 2018)。
Nuclear dynamics of migratory granule cells
移動する顆粒細胞の核動態
培養下で移動する顆粒細胞の核(H2B:マゼンダ、白)と微小管(Dcx; 緑)を高時空間解像タイムラプスにより観察した。
現在、移動するニューロンにかかる「力」の実体を探索しています。高解像顕微鏡イメージ解析、微小応力分布の定量解析、分子生物学手法を組み合わせた融合的アプローチにより、動く細胞に負荷される力を推定し、その分子実体の解明を目指しています。
Reference
Wu YK, Umeshima H, Kurisu J and Kengaku M. “Nesprins and opposing microtubule motors generate a point force that drives directional nuclear motion in migrating neurons.”
Development, 145, 10.1242/dev.158782. (2018)
Nakashima K, Umeshima H and Kengaku M. “Cerebellar granule cells are predominantly generated by terminal symmetric divisions of granule cell precursors.”
Dev Dyn, 244(6), 748-758 (2015)
Umeshima H and Kengaku M. “Differential roles of cyclin-dependent kinase 5 in tangential and radial migration of cerebellar granule cells.”
Mol Cell Neurosci, 52, 62-72 (2013)
Umeshima H, Hirano T and Kengaku M. “Microtubule-based nuclear movement occurs independently of cetrosome positioning in migrating neurons.”
Proc Natl Acad Sci U S A, 104, 16182-16187 (2007) Must Read Article by Faculty of 1000
Development of techniques
We seek to develop new technologies to analyze and manipulate the dynamic cell motility during brain development. We introduce cross-disciplinary approaches such as mechanobiology, advanced microscopy, and materials chemistry to analyze phenomena that were previously invisible. The following list is a part of our research activities.
- Traction force microscopy of migrating neurons (Umeshima et al, 2018)
- Identification of cytoskeletal organization in neurons by super-resolution microscopy (Fujishima et al., 2020)
- Reconstruction of 3D cell architectures of the brain using artificial scaffolds (Fujishima et al., 2020)
- Gene-switching using gold nano-rods (Nakatsuji et al., Sci Rep 2017)
イメージングを用いた新規解析系の開拓
所属するiCeMSの解析センターのイメージング設備を利用し、発生脳における動的な細胞運動を観察・解析するライブイメージング技術の開拓を行っています。また、メカノバイオロジー、先端光学顕微鏡、材料化学などの分野融合的アプローチにより、これまで不可能だった解析に取り組んでいます。以下は現在までに取り組んできたテーマの一部です。
- ニューロン移動の牽引力顕微鏡解析 (Umeshima et al, 2018)
- ニューロン細胞骨格の超解像顕微鏡解析 (Fujishima et al., 2020)
- 人工スキャフォールドを用いた脳皮質のin vitro再構築 (Fujishima et al., 2020)
- 金ナノロッドを用いた遺伝子発現制御 (Nakatsuji et al., Sci Rep 2017)
Reference
Fujishima K, Kurisu J, Yamada M, Kengaku M. “βIII spectrin controls the planarity of Purkinje cell dendrites by modulating perpendicular axon-dendrite interactions.”
Development. 2020 Dec 16;147(24):dev194530. doi: 10.1242/dev.194530.
Umeshima H, Nomura KI, Yoshikawa S, Hörning M, Tanaka M, Sakuma S, Arai F, Kaneko M and Kengaku M. “Local traction force in the proximal leading process triggers nuclear translocation during neuronal migration.”
Neurosci Res, 10.1016/j.neures.2018.04.001. (2018)
Nakatsuji H, Kawabata Galbraith K, Kurisu J, Imahori H, Murakami T and Kengaku M. “Surface chemistry for cytosolic gene delivery and photothermal transgene expression by gold nanorods.”
Sci. Rep, 7:4694 (2017)